Monthly Botox & Dermal Filler Training Certification - Austin, Texas 2020 Middletown United States
- Cosmetic Medical Training
- Middletown Delaware
- United States
- Aesthetic
Monthly Botox & Dermal Filler Training Certification - Austin, Texas 2020 Middletown United States
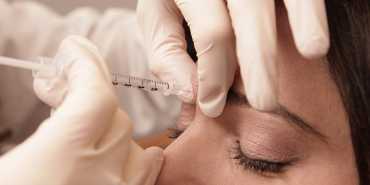
Our Austin, Texas Trainer will come to your office to train you on how to inject Botox® and Dermal Fillers during any day this month.
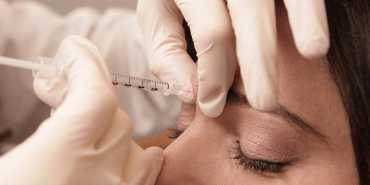
Read MoreMonthly Botox & Dermal Filler Training Certification - Indianapolis, Indiana 2020 Fremont United States
- Cosmetic Medical Training
- Fremont California
- United States
- Aesthetic
Monthly Botox & Dermal Filler Training Certification - Indianapolis, Indiana 2020 Fremont United States
Read MoreThe new age for healthcare IT: How is digital healthcare transforming the industry? 2020 Fremont United States
- Archer Software
- Fremont California
- United States
- Medical Technology, Orthopedics, Surgery, Healthcare Management, Mental health or psychiatric nursing, Hospital Management, Nursing management
The new age for healthcare IT: How is digital healthcare transforming the industry? 2020 Fremont United States
Read MoreInternational Conference on Nursing & Healthcare 2020 Prague Czechia
- Alisha Williams
- Prague
- Czechia
- Advanced practice registered nursing, Healthcare Management, Public Health, Critical care nursing
International Conference on Nursing & Healthcare 2020 Prague Czechia
Read MoreInternational Conference on Surgery & Plastic Surgery 2020 Prague Czechia
- Alisha Williams
- Prague
- Czechia
- Surgery, Plastic Surgery, Cardiovascular Surgery, Colon and Rectal Surgery
International Conference on Surgery & Plastic Surgery 2020 Prague Czechia
Read More30th International Conference on Pediatrics & Primary Care 2020 London United Kingdom
- Conference Series llc LTD
- London
- United Kingdom
- General/Internal Med
30th International Conference on Pediatrics & Primary Care 2020 London United Kingdom
Read More11th International Conference on Alternative Medicine and Holistic Medicine 2020 Zurich Switzerland
- Euroscicon ltd
- Zurich
- Switzerland
- Holistic Medicine, Public Health
11th International Conference on Alternative Medicine and Holistic Medicine 2020 Zurich Switzerland
Read More5th World Online Conference on Pediatrics 2020
5th World Online Conference on Pediatrics 2020
Read MoreDigital transformation in medical education 2020 Middletown United States
- UpHill Health
- Middletown Delaware
- United States
- Medical Technology, Healthcare Management
Digital transformation in medical education 2020 Middletown United States
Digital transformation in medical education
Read More24th Edition of International Conference on World Cardiology 2020 Zurich Switzerland
- Euroscicon
- Zurich
- Switzerland
- Cardiology
24th Edition of International Conference on World Cardiology 2020 Zurich Switzerland
Read MoreInfectious Diseases Virtual 2020 2020 Middletown United States
- Magnus Group
- Middletown Delaware
- United States
- Infectious Diseases
Infectious Diseases Virtual 2020 2020 Middletown United States
Read Morecardiology 2020 New York United States
- John Benson
- New York New York
- United States
- Biomedical Engineering
cardiology 2020 New York United States
Read More3rd World Heart Rhythm Conference 2020 Cape Town South Africa
- ME Conferences
- Cape Town
- South Africa
- General Clinical Cardiology, Nuclear Cardiology, Cardiology
3rd World Heart Rhythm Conference 2020 Cape Town South Africa
Read More2nd World conference on Vaccine and Immunology in Helsinki, Finland
- ME Conferences
- Helsinki
- Finland
- Pathology
2nd World Conference on Vaccine and Immunology in Helsinki, Finland
Read More16th European Congress on Clinical Pediatrics and Child Care 2020 Budapest Hungary
- Conference Series
- Budapest
- Hungary
- Pediatrics
16th European Congress on Clinical Pediatrics and Child Care 2020 Budapest Hungary
Dear Colleague,
Read More
16th European Congress on Clinical Pediatrics and Child Care 2020 Budapest Hungary
- Conference Series
- Budapest
- Hungary
- Pediatric nursing
16th European Congress on Clinical Pediatrics and Child Care 2020 Budapest Hungary
Dear Colleague,
Read More
Webinar on Pharmacology 2020 Paris France
- Meetings International Pte Ltd
- Paris
- France
- Pharmaceutical
Webinar onPharmacology 2020 Paris France
Read More19th International Conference on Gastroenterology and Digestive Disorders 2020 Abu Dhabi United Arab Emirates
- ME Conference
- Abu Dhabi
- United Arab Emirates
- Gastroenterology
19th International Conference on Gastroenterology and Digestive Disorders 2020 Abu Dhabi UAE United Arab Emirates
Read More3rd EuroSciCon Congress on Heart Disease & Interventional Cardiology 2020 Edinburgh Suriname
- Euroscicon ltd
- Edinburgh
- Suriname
- Cardiology
3rd EuroSciCon Congress on Heart Disease & Interventional Cardiology
Read More19th International Conference on Gastroenterology and Digestive Disorders 2020 United Arab Emirates
- ME Conferences
- United Arab Emirates
- Gastroenterology
19th International Conference on Gastroenterology and Digestive Disorders
Read More8th International Conference on Brain Disorder and Therapeutics 2020 Barcelona Spain
- Allied Academies
- Barcelona Barcelona
- Spain
- Neurology, Psychology, Academic – CME
CME & CPD Accredited 8th International Conference on Brain Disorder and Therapeutics 2020 Barcelona Spain
Read More2nd World Brain Congress 2020 Dubai United Arab Emirates
- ME Conferences
- Dubai
- United Arab Emirates
- Academic – CME, Cardiology, Cardiac nursing, Cardiovascular Surgery
2nd World Brain Congress 2020 Dubai United Arab Emirates
Read MoreWorld Congress on Surgeons 2020 Vienna Austria
- Euroscicon
- Vienna
- Austria
- General Surgery
World Congress on Surgeons 2020 Vienna Austria
Greetings of the Day!
Read More11th International Conference on New Frontiers in Clinical Microbiology and Infectious Diseases 2020 London UK
- Conferenceseries LLC LTD
- Bath Road, Heathrow, London Heathrow, UB7 0DU London United Kingdom
- United Kingdom
- Academic – CME
11th International Conference on New Frontiers in Clinical Microbiology and Infectious Diseases 2020 Bath Road, Heathrow, London Heathrow, UB7 0DU London United Kingdom United Kingdom
Read MoreHealth and Medical Tourism 2020 United Arab Emirates
- Longdom Group
- United Arab Emirates
- Academic – CME
Health and Medical Tourism 2020 United Arab Emirates
Read MoreCopyright © 2015 - 2121 World Medical Events Organization - All Rights Reserved | Terms & Conditions | Privacy Policy